 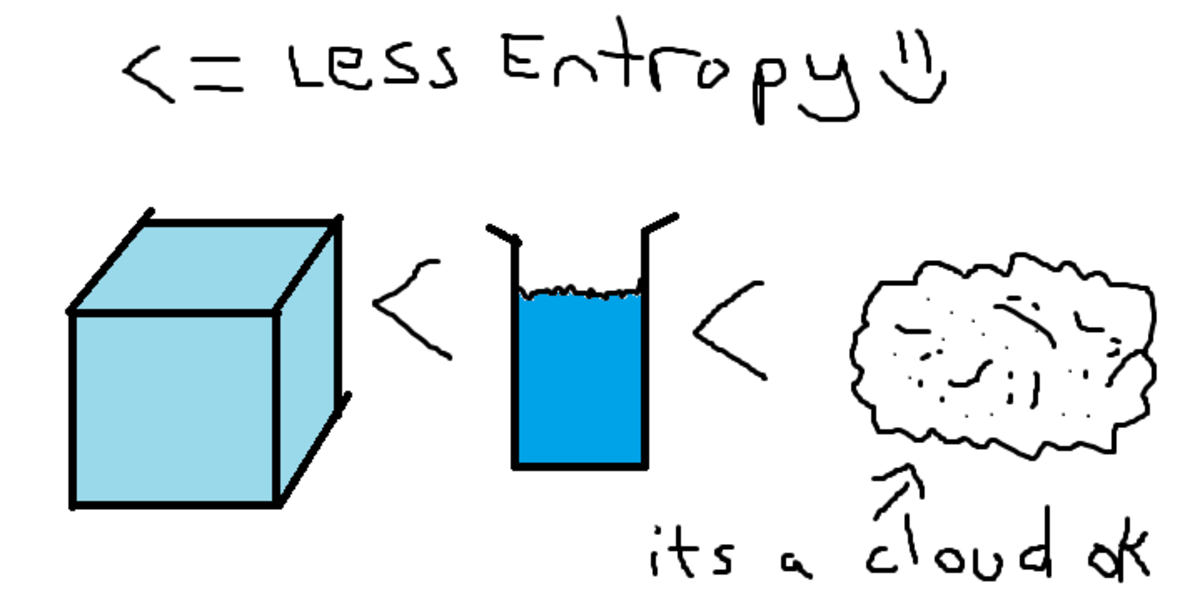
It follows that any perturbation of the system, whether it is a random jiggling of its parts or a whack from the outside, will, by the laws of probability, nudge the system toward disorder or uselessness. Now order could be characterized in terms of the set of all microscopically distinct states of a system: Of all these states, the ones that we find useful make up a tiny sliver of the possibilities, while the disorderly or useless states make up the vast majority. Once it was appreciated that heat is not an invisible fluid but the motion of molecules, a more general, statistical version of the Second Law took shape. In its original formulation the Second Law referred to the process in which usable energy in the form of a difference in temperature between two bodies is dissipated as heat flows from the warmer to the cooler body. (The First Law is that energy is conserved the Third, that a temperature of absolute zero is unreachable.) Closed systems inexorably become less structured, less organized, less able to accomplish interesting and useful outcomes, until they slide into an equilibrium of gray, tepid, homogeneous monotony and stay there. For an isolated system, the natural course of events takes the system to a more disordered (higher entropy) state.The Second Law of Thermodynamics states that in an isolated system (one that is not taking in energy), entropy never decreases. If snapshots of a system at two different times shows one state which is more disordered, then it could be implied that this state came later in time. 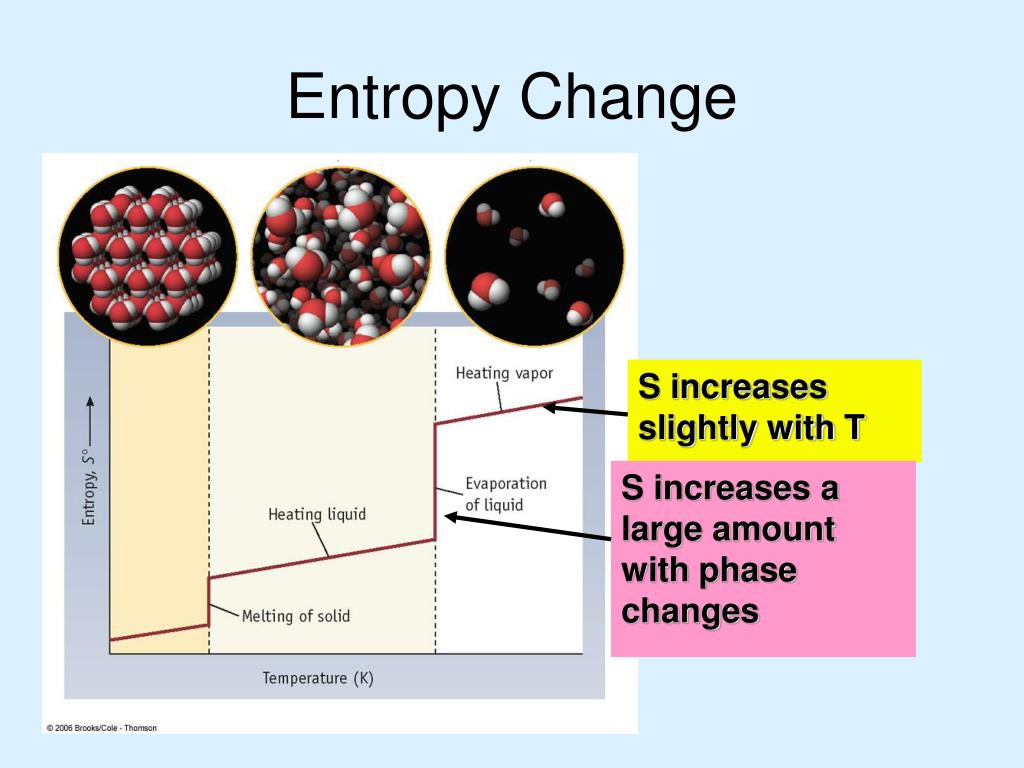
Since entropy gives information about the evolution of an isolated system with time, it is said to give us the direction of " time's arrow". Entropy:Ī state variable whose change is defined for a reversible process at T where Q is the heat absorbed.Ī measure of the amount of energy which is unavailable to do work.Ī measure of the multiplicity of a system. Second Law of Thermodynamics: In any cyclic process the entropy will either increase or remain the same. Alternative statements: Second Law of Thermodynamics Work is required to transfer net energy to the hot object. Energy can transfer from the cold object to the hot object either by transfer of energetic particles or electromagnetic radiation, but the net transfer will be from the hot object to the cold object in any spontaneous process. It is important to note that when it is stated that energy will not spontaneously flow from a cold object to a hot object, that statement is referring to net transfer of energy. This is the "second form" or Clausius statement of the second law. The statements about refrigerators apply to air conditioners and heat pumps, which embody the same principles. 
Energy will not flow spontaneously from a low temperature object to a higher temperature object. Second Law of Thermodynamics: It is not possible for heat to flow from a colder body to a warmer body without any work having been done to accomplish this flow. This is sometimes called the "first form" of the second law, and isreferred to as the Kelvin-Planck statement of the second law. Some amount of heat Q C must be exhausted to a cold reservoir. Second Law of Thermodynamics: It is impossible to extract an amountof heat Q H from a hot reservoir and use it all to do work W. Qualitative statements of the Second Law of Thermodynamics The maximum efficiency which can be achieved is the Carnot efficiency. Its implications may be visualized in terms of the waterfall analogy. In so doing, it goes beyond the limitations imposed by the first law of thermodynamics. The second law of thermodynamics is a general principle which places constraints upon the direction of heat transfer and the attainable efficiencies of heat engines. Second Law of Thermodynamics Second Law of Thermodynamics 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
